Basics of UV-visible absorption spectroscopy
Absorption spectrum
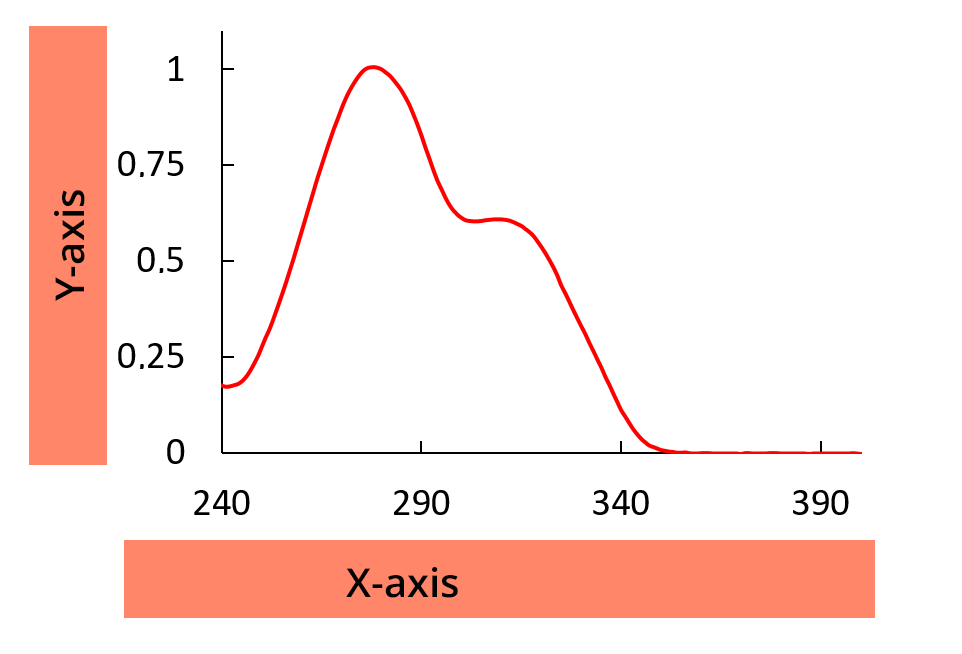
An absorption spectrum represents the absorption of a molecule as a function of wavelength.
Y-axis
Y-axis = absorbance A or transmittance T
Aλ = log I0/I without unit
Tλ = 10-Aλ = I/I0
X-axis
On the X-axis = wavelength in nm
Click on each box to find out more
Chemical groups responsible for absorption are called chromophores.
For gases, absorption bands are thin, although for liquids they can be tens of nanometers wide.