Determination of NO3- ions with a selective electrode (potentiometry)
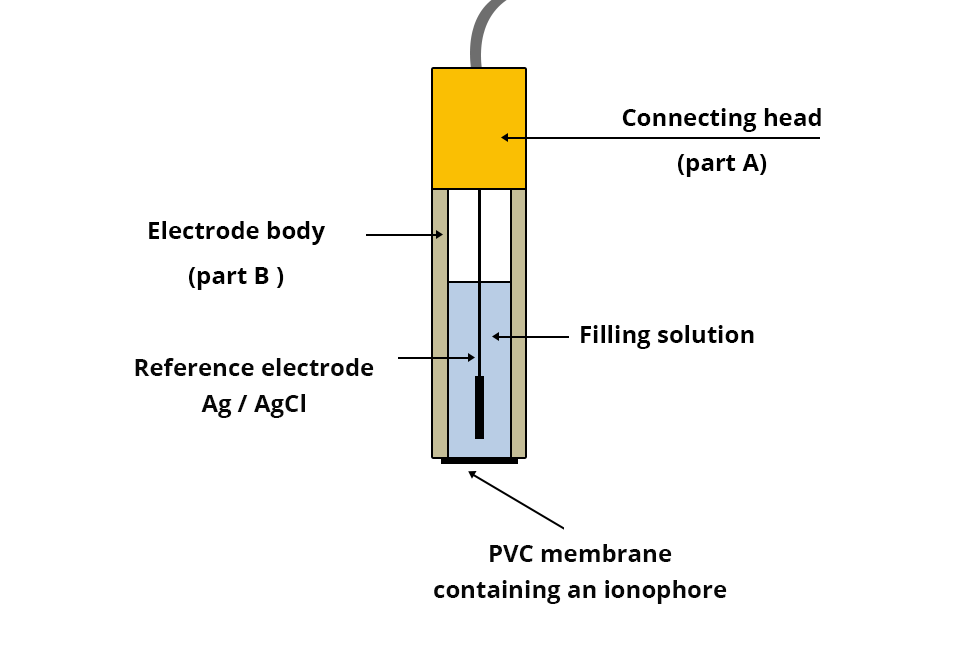
Constitution of the electrode
As with the calcium ion selective electrode, this electrode consists of:
- an Ag/AgCl reference electrode (chloride silver wire) connected via a connecting head (part A) to a conducting wire;
- A plastic body containing at its end a PVC (polyvinyl chloride) membrane containing an ionophore capable of almost selectively complexing the nitrate ions NO3- (active part of the electrode);
- An internal KNO3 filling solution supplied by the manufacturer.