Preparing a dilution

Water is not always the best solvent for preparing a solution! Be sure to choose the right solvent!

Side note: this video was made to emphasize the important practical aspects, while at the same time keeping it humorous.
Introducing a solution with a graduated pipette
The volume introduced must correspond to the nominal volume of the pipette
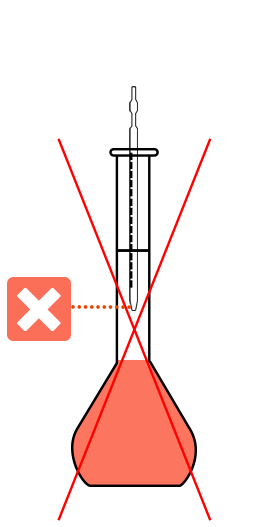
No contact between the pipette and the side of the flask.
No capillary action for the last drops
The poured volume is not correct
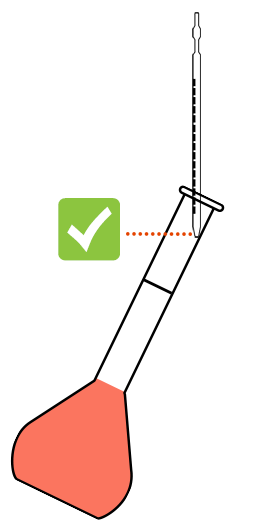
Contact between the pipette and the side of the flask.
Suction of the last drops by capillary action
The poured volume is correct

Never withdraw the stock solution directly (solution of known concentration or commercial solution) from its container. It is imperative to use an intermediate container.





