ON WHICH PRINCIPLE?
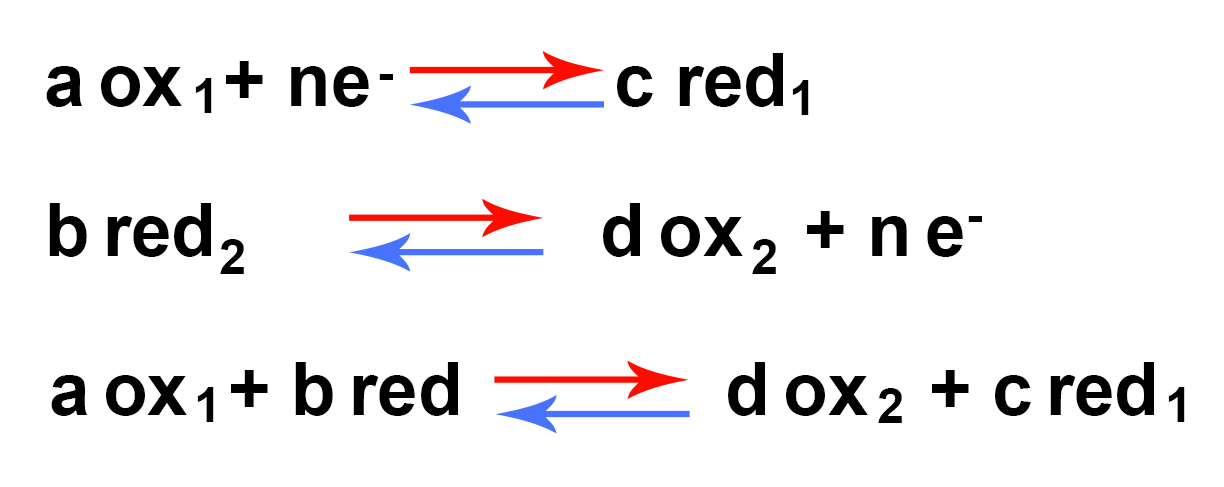
Redox couple:
Redox reaction involves the concept of a redox couple.
To be able to fix one or more electrons, any oxidizer needs a reductant that can transfer them. The transfer of electrons takes place between two redox couples, one of which oxidizes while the other reduces. There can be no oxidation without a simultaneous reduction, and the other way around.
The couple with the highest redox potential is the oxidizer.