Determination of SO42- ions by conductimetric titration
Procedure
- Sample 50 mL of the water to be analyzed and transfer to a 500 mL beaker.
- Add 200 mL of distilled water.
- Stir.
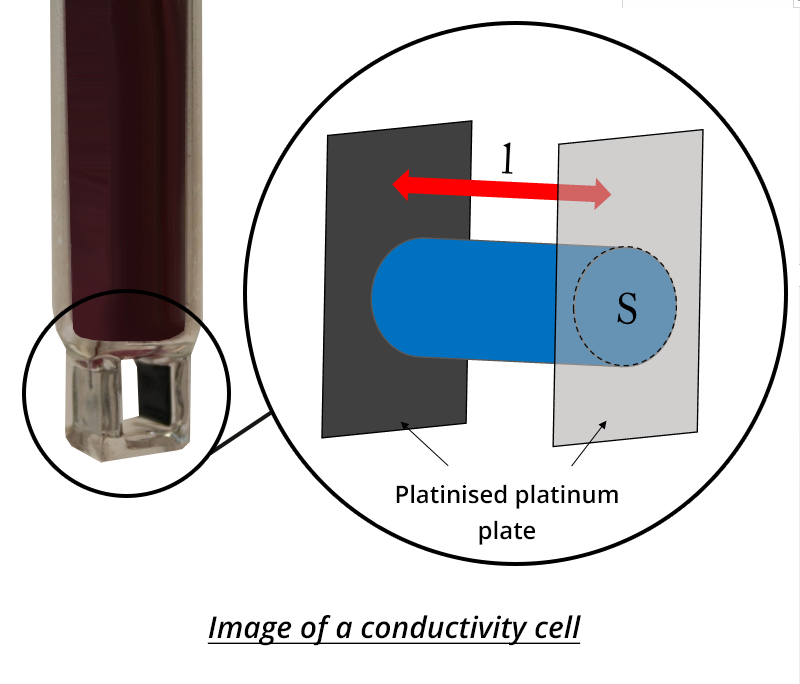
- Place the conductimetric cell in the solution.
- Dose with a barium chloride solution at 0.05 mol/L.
- The conductivity σ is determined from the volume of barium chloride solution added.
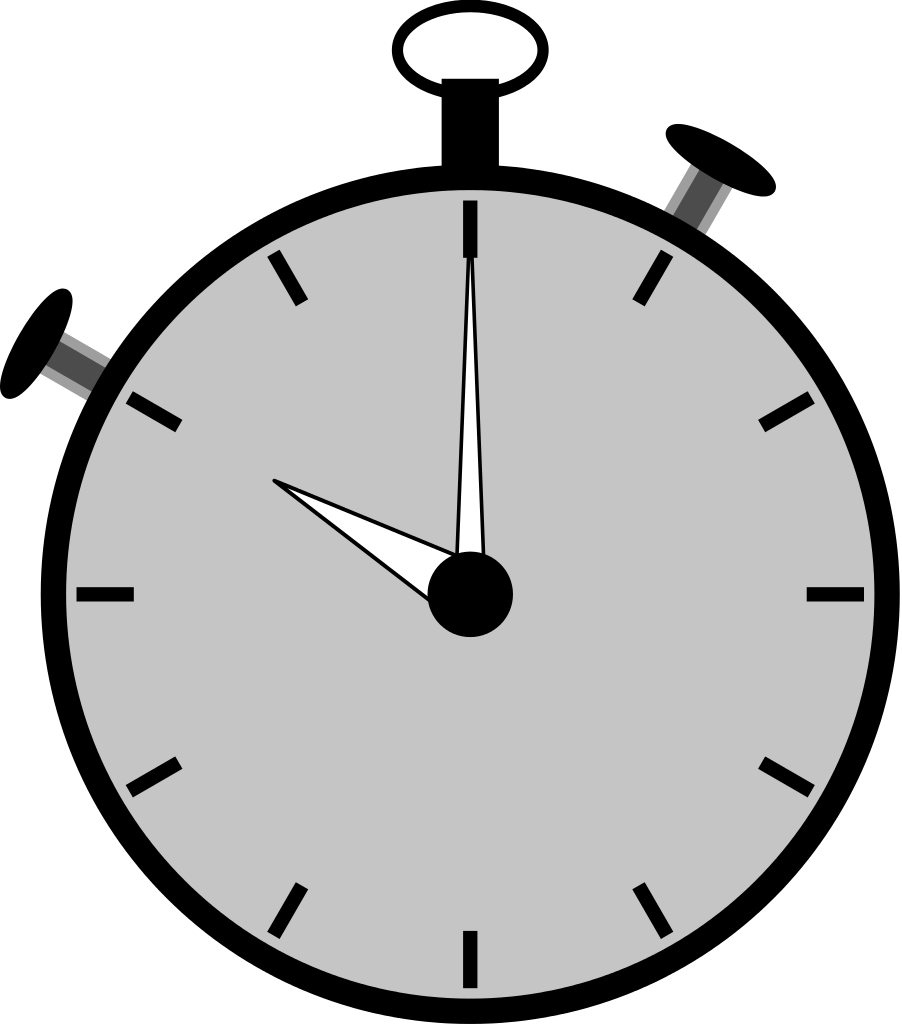 ≈ 30 minutes / sample
≈ 30 minutes / sample