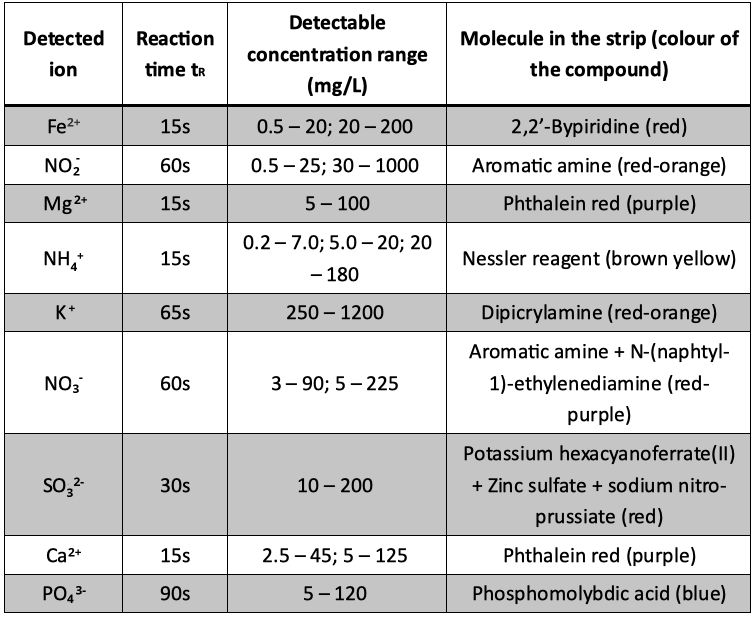
Analysis of a spring water
Reflectometry
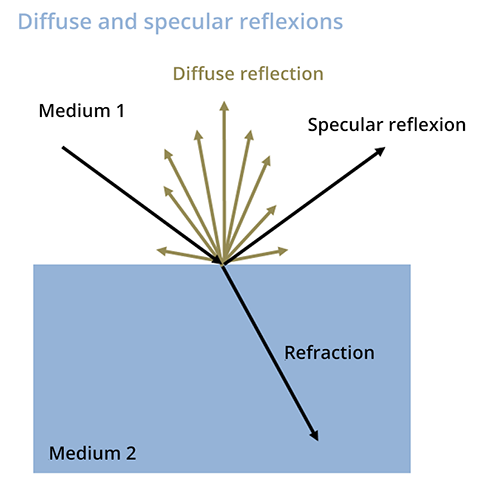
This technique consists in assessing the intensity of the diffuse and specular reflection of a sample. One of the components is a strip, containing a molecule capable of selectively complexing or reacting with a molecule of interest to form a colored compound.
This selective strip is dipped into the solution we wish to analyze for two seconds. A reaction occurs on the strip, with a reaction time, tReaction, and colors it. The coloring of the strip is all the more intense as the ion of interest in solution is present in high concentration. Before placing the strip in the reflectometer, the excess liquid is removed with absorbent paper.

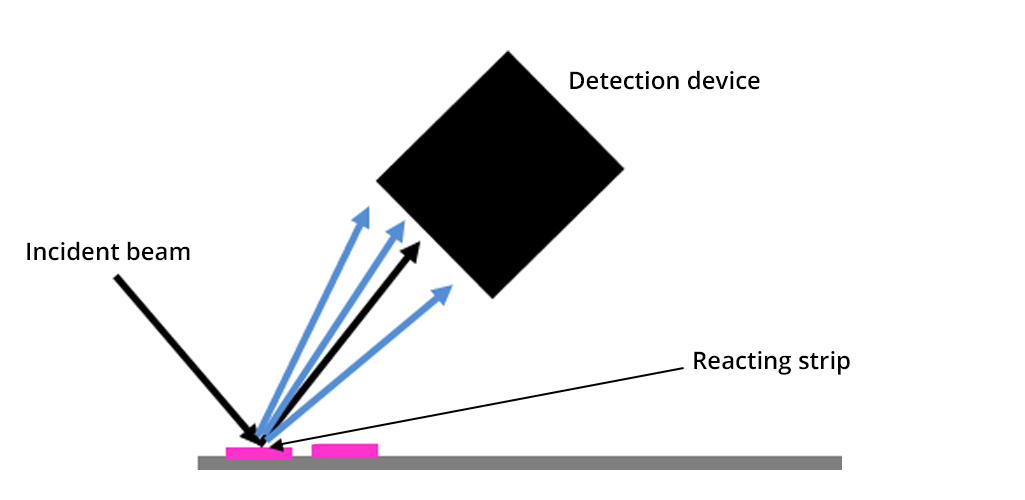
For each detected ion, it is necessary to calibrate the detection device before the experiment, with a transparent strip specific to each ion. This step allows the detection device to calibrate the diffusion intensity according to the ion concentration to be analyzed. The concentration is therefore determined through a quantitative internal color scale.